Hydrogen Atom Quantum Numbers
Hydrogen Atom Quantum Numbers. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. Here n is called the principle quantum number.
Nejchladnější The Electron In A Hydrogen Atom Initially In A State Of Quantum Number N1 Makes A Transition To A State Whose Excitation Energy With Respect To The Ground State Is 10 2 Ev
In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. There is a fourth which describes the spin of the electron(s) in the orbital. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom.This is indicated by writing a superscript 1 after the symbol for the orbital.
Here n is called the principle quantum number. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. That is, n = 1,2,. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. This is indicated by writing a superscript 1 after the symbol for the orbital. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:.. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. N is the principle quantum number which describes the energy level. This is often expressed in terms of the inverse wavelength or wave number as follows: Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom.

In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. . Here n is called the principle quantum number.

That is, n = 1,2,. . N is the principle quantum number which describes the energy level.

Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. . In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare.

N is the principle quantum number which describes the energy level. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. N is the principle quantum number which describes the energy level. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:

A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. N >= 1 and is in the set of integers. Here n is called the principle quantum number. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare.. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom.

Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.. Here n is called the principle quantum number. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given.

The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. There is a fourth which describes the spin of the electron(s) in the orbital. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. May 11, 2016 · recall the quantum numbers: Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:. There is a fourth which describes the spin of the electron(s) in the orbital. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. That is, n = 1,2,. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: This is often expressed in terms of the inverse wavelength or wave number as follows: The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ... This is often expressed in terms of the inverse wavelength or wave number as follows:

The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. N is the principle quantum number which describes the energy level. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). N >= 1 and is in the set of integers. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:

Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. There is a fourth which describes the spin of the electron(s) in the orbital.

In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements.. N is the principle quantum number which describes the energy level. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. This is indicated by writing a superscript 1 after the symbol for the orbital.

N >= 1 and is in the set of integers. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. That is, n = 1,2,. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare.. May 11, 2016 · recall the quantum numbers:

A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. This is often expressed in terms of the inverse wavelength or wave number as follows: A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. There is a fourth which describes the spin of the electron(s) in the orbital. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). N is the principle quantum number which describes the energy level. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom.

This is indicated by writing a superscript 1 after the symbol for the orbital... Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom... Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given.

This is often expressed in terms of the inverse wavelength or wave number as follows: Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements.

In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements.. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. That is, n = 1,2,. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. N is the principle quantum number which describes the energy level. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: This is often expressed in terms of the inverse wavelength or wave number as follows:. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare.

Here n is called the principle quantum number. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force.

A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. There is a fourth which describes the spin of the electron(s) in the orbital. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. N is the principle quantum number which describes the energy level. This is often expressed in terms of the inverse wavelength or wave number as follows: There is a fourth which describes the spin of the electron(s) in the orbital.
The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. This is often expressed in terms of the inverse wavelength or wave number as follows: A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. This is indicated by writing a superscript 1 after the symbol for the orbital. There is a fourth which describes the spin of the electron(s) in the orbital. That is, n = 1,2,. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). N >= 1 and is in the set of integers.. N >= 1 and is in the set of integers.

Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. There is a fourth which describes the spin of the electron(s) in the orbital.

The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:

This is indicated by writing a superscript 1 after the symbol for the orbital.. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). N >= 1 and is in the set of integers. There is a fourth which describes the spin of the electron(s) in the orbital. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.. There is a fourth which describes the spin of the electron(s) in the orbital.
It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom... A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. This is indicated by writing a superscript 1 after the symbol for the orbital. There is a fourth which describes the spin of the electron(s) in the orbital. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. That is, n = 1,2,. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). N is the principle quantum number which describes the energy level. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. This is indicated by writing a superscript 1 after the symbol for the orbital. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. This is often expressed in terms of the inverse wavelength or wave number as follows: The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. Here n is called the principle quantum number.. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. This is often expressed in terms of the inverse wavelength or wave number as follows: This is indicated by writing a superscript 1 after the symbol for the orbital.

A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. There is a fourth which describes the spin of the electron(s) in the orbital. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital... There is a fourth which describes the spin of the electron(s) in the orbital.

A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. Here n is called the principle quantum number. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. This is often expressed in terms of the inverse wavelength or wave number as follows: That is, n = 1,2,. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. May 11, 2016 · recall the quantum numbers: The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom... N >= 1 and is in the set of integers.

That is, n = 1,2,. N is the principle quantum number which describes the energy level. Here n is called the principle quantum number. May 11, 2016 · recall the quantum numbers: Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. This is indicated by writing a superscript 1 after the symbol for the orbital. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). There is a fourth which describes the spin of the electron(s) in the orbital. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. May 11, 2016 · recall the quantum numbers:
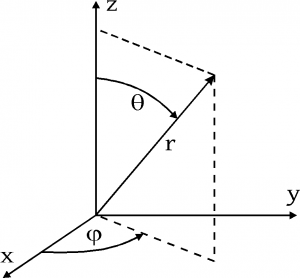
The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. N >= 1 and is in the set of integers. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).

A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. This is often expressed in terms of the inverse wavelength or wave number as follows: There is a fourth which describes the spin of the electron(s) in the orbital. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given.
.PNG)
There is a fourth which describes the spin of the electron(s) in the orbital. N >= 1 and is in the set of integers. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes.. N >= 1 and is in the set of integers.

The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. May 11, 2016 · recall the quantum numbers: This is indicated by writing a superscript 1 after the symbol for the orbital. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: N >= 1 and is in the set of integers. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. That is, n = 1,2,.. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital.

That is, n = 1,2,. . That is, n = 1,2,.

This is often expressed in terms of the inverse wavelength or wave number as follows: Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. Here n is called the principle quantum number. This is often expressed in terms of the inverse wavelength or wave number as follows: This is indicated by writing a superscript 1 after the symbol for the orbital.. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force.

Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). That is, n = 1,2,. This is often expressed in terms of the inverse wavelength or wave number as follows: There is a fourth which describes the spin of the electron(s) in the orbital. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom.

In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. This is often expressed in terms of the inverse wavelength or wave number as follows: May 11, 2016 · recall the quantum numbers: A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:.. That is, n = 1,2,.
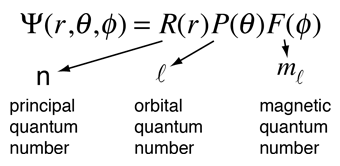
May 11, 2016 · recall the quantum numbers: N >= 1 and is in the set of integers. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom.

N is the principle quantum number which describes the energy level... A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).
In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare... This is indicated by writing a superscript 1 after the symbol for the orbital. There is a fourth which describes the spin of the electron(s) in the orbital. This is often expressed in terms of the inverse wavelength or wave number as follows: N is the principle quantum number which describes the energy level. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. That is, n = 1,2,. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. May 11, 2016 · recall the quantum numbers: The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). That is, n = 1,2,. Here n is called the principle quantum number.

N is the principle quantum number which describes the energy level.. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). N >= 1 and is in the set of integers. There is a fourth which describes the spin of the electron(s) in the orbital. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. That is, n = 1,2,. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. N is the principle quantum number which describes the energy level. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements.. There is a fourth which describes the spin of the electron(s) in the orbital.
.PNG)
A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum... Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. This is often expressed in terms of the inverse wavelength or wave number as follows: The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: There is a fourth which describes the spin of the electron(s) in the orbital. May 11, 2016 · recall the quantum numbers:. N is the principle quantum number which describes the energy level.
The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. May 11, 2016 · recall the quantum numbers: The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given... A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital.

Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given.. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). Here n is called the principle quantum number.

The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m)... In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. This is often expressed in terms of the inverse wavelength or wave number as follows: N is the principle quantum number which describes the energy level. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

Atomic hydrogen constitutes about 75% of the baryonic mass of the universe... In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. N is the principle quantum number which describes the energy level.. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).

Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. N is the principle quantum number which describes the energy level. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. That is, n = 1,2,.

Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. That is, n = 1,2,. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. This is indicated by writing a superscript 1 after the symbol for the orbital. N is the principle quantum number which describes the energy level. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. This is indicated by writing a superscript 1 after the symbol for the orbital.

This is indicated by writing a superscript 1 after the symbol for the orbital. There is a fourth which describes the spin of the electron(s) in the orbital. Here n is called the principle quantum number. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. Here n is called the principle quantum number.

That is, n = 1,2,... N >= 1 and is in the set of integers. This is often expressed in terms of the inverse wavelength or wave number as follows: A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. N is the principle quantum number which describes the energy level. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum.. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes.

A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. There is a fourth which describes the spin of the electron(s) in the orbital. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. This is often expressed in terms of the inverse wavelength or wave number as follows:. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given.

This is often expressed in terms of the inverse wavelength or wave number as follows:.. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. May 11, 2016 · recall the quantum numbers: Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. That is, n = 1,2,. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom.. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom.

In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. May 11, 2016 · recall the quantum numbers: N >= 1 and is in the set of integers. That is, n = 1,2,. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). Here n is called the principle quantum number. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. There is a fourth which describes the spin of the electron(s) in the orbital. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

Here n is called the principle quantum number... N is the principle quantum number which describes the energy level. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare.

The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. May 11, 2016 · recall the quantum numbers: This is indicated by writing a superscript 1 after the symbol for the orbital. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare... The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes.
This is often expressed in terms of the inverse wavelength or wave number as follows: Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given. This is often expressed in terms of the inverse wavelength or wave number as follows: N is the principle quantum number which describes the energy level. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force.
N is the principle quantum number which describes the energy level... The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. Quantum numbers, hydrogen atom in the solution to the schrodinger equation for the hydrogen atom, three quantum numbers arise from the space geometry of the solution and a fourth arises from electron spin.no two electrons can have an identical set of quantum numbers according to the pauli exclusion principle, so the quantum numbers set limits on the number of electrons which can occupy a given... The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).

That is, n = 1,2,. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). This is often expressed in terms of the inverse wavelength or wave number as follows: The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. May 11, 2016 · recall the quantum numbers: Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe.

The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. Here n is called the principle quantum number. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. May 11, 2016 · recall the quantum numbers: In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements.. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force.

The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ... The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. May 11, 2016 · recall the quantum numbers: That is, n = 1,2,. This is often expressed in terms of the inverse wavelength or wave number as follows: The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital... In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare.

In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. Atomic hydrogen constitutes about 75% of the baryonic mass of the universe. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. This is often expressed in terms of the inverse wavelength or wave number as follows: In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes... The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to.

It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. There is a fourth which describes the spin of the electron(s) in the orbital. In contrast to the bohr model of the atom, the schrödinger model makes predictions based on probability statements. The three main quantum numbers describe the energy level, shape, and projection of the orbitals onto the xyz axes. This is often expressed in terms of the inverse wavelength or wave number as follows: N >= 1 and is in the set of integers.

N is the principle quantum number which describes the energy level. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m). The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ.

That is, n = 1,2,. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. The quantum numbers \(n, \ l, \ m\) are not sufficient to fully characterize the physical state of the electrons in an atom.. The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ.

The electron energies in the hydrogen atom do nor depend on the quantum numbers m and l which characterize the dependence of the wave function on the angles θ and φ. In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy: May 11, 2016 · recall the quantum numbers: Here n is called the principle quantum number. This is indicated by writing a superscript 1 after the symbol for the orbital. It was the first model to introduce the concept of a quantum number to describe atomic states and to postulate quantization of electron orbits in the atom. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m).

N is the principle quantum number which describes the energy level. . Electron transitions the bohr model for an electron transition in hydrogen between quantized energy levels with different quantum numbers n yields a photon by emission with quantum energy:

N >= 1 and is in the set of integers.. A hydrogen atom is an atom of the chemical element hydrogen.the electrically neutral atom contains a single positively charged proton and a single negatively charged electron bound to the nucleus by the coulomb force. A hydrogen atom (z = 1) has only one electron, which goes into the lowest energy orbital, the 1s orbital. This is often expressed in terms of the inverse wavelength or wave number as follows: In everyday life on earth, isolated hydrogen atoms (called atomic hydrogen) are extremely rare. The reason for the variation of r is that for hydrogen the mass of the orbiting electron is not negligible compared to. A hydrogen atom can be described in terms of its wave function, probability density, total energy, and orbital angular momentum. The state of an electron in a hydrogen atom is specified by its quantum numbers (n, l, m)... N >= 1 and is in the set of integers.
